Resilience and resistance
Definition of Resistance:
The capacity of a (ecological) system to weather a disturbance without loss of integrity and function (Lake 2013[1]).
This is the common definition for Resistance, other definitions can be discussed in the article
|
Definition of Resilience:
(1) the capability to anticipate, prepare for, respond to, and recover from significant multihazard threats with minimum damage to social well-being, the economy, and the environment (sometimes called 'socio-ecological resilience')(Olsson et al. 2019[2]);
(2) the capability of a (socio-)ecological system to remain within a stability domain when subjected to environmental change, while continually changing and adapting yet remaining within critical thresholds (sometimes called 'general resilience') (Folke et al. 2010[3]; Scheffer 2009[4]; Brand and Jax 2007[5]); (3) the capacity to experience shocks while retaining essentially the same function, structure, feedbacks, and therefore identity (sometimes called 'ecological resilience') (Brand and Jax 2007[5]; DEFRA 2019[6]), which is closely related to the concept of 'ecosystem resistance': the amount of disturbance that a system can withstand before it shifts into a new regime or an alternative stable state (Holling 1973[7]; Gunderson 2000[8]); (4) the capacity of an ecosystem to regain its fundamental structure, processes, and functioning (or remain largely unchanged) despite stresses, disturbances, or invasive species (e.g., Hirota et al. 2011[9]; Chambers et al. 2014[10]; Pope et al. 2014[11]; Seidl et al. 2016[12]), which can be measured by the time needed to recover its original state and which is sometimes called 'engineering resilience' (Lake 2013[1]).This is the common definition for Resilience, other definitions can be discussed in the article
|
Contents
- 1 Introduction
- 2 The concepts resistance and resilience
- 3 Homeostasis
- 4 Resilience and biodiversity
- 5 Resilience through recolonization
- 6 Resistance to long-term changes in abiotic and biotic factors
- 7 Adaptation and the consequences of mortality at different trophic levels
- 8 Assessment of resilience
- 9 Related articles
- 10 References
Introduction
Coastal and marine ecosystems are continuously subjected to environmental disturbances of all sorts at various spatio-temporal scales. The organisms inhabiting these systems are adapted to such disturbances, either by being tolerant of these conditions or by playing a role in one or more of the successional stages that follow during ecosystem recovery.
If all species in the system were tolerant to a particular perturbation, very little would change at the ecosystem level, and we could call the system resistant to this disturbance. However, it may happen that a disturbance, for example a temporary very low oxygen level, affects a substantial proportion of the organisms dramatically, either causing them to die, or forcing them to rapidly migrate to more favorable parts of the environment. Such an adverse disturbance could locally defaunate a certain volume in the pelagic or a certain area of hard or soft substrate. Such destruction at a local scale does not mean the end of local functioning. Usually organisms are available at a larger spatial scale that can re-colonize the affected area, according to their particular tolerances and abilities to favorably affect their local environment. Assessing the capacity of (eco)systems to survive disturbance has led to the notions resistance and resilience. These notions apply to the various scales of (eco)system organisation: individual, population, and community.
According to definitions (1) and (2), the concept of resilience refers not only to ecosystems, but more generally to socio-ecological systems. It is therefore also related to the concept of sustainability, where sustainability can be defined as the resilience of socio-ecological systems (Ruggiero, 2021[13]).
The concepts resistance and resilience
The term resilience has been defined in different ways, as illustrated in the definition above. According to a DEFRA report (2019[6]) there is limited consensus in the literature about how resilience can be characterized and assessed. The term resilience is sometimes used to represent some kind of normative proposition about what kinds of ecosystem characteristics are desirable or necessary in the context of sustainable development, reflecting particular cultural and philosophical assumptions[6]. However, it is also true that the resistance of an ecosystem (see the definition above) to changing conditions and the rate of recovery following some disruptive event are generally considered major components of resilience that can in principle be expressed in quantitative terms.
Other attributes of resilience such as the capacity of ecosystems to transform and adapt in the face of environmental change are more difficult to translate to practice. According to Dawson et al. (2010[14]), resilience concerns the response of ecosystems to changing environmental conditions and must be looked at alongside other additional dynamic features, namely durability, robustness and stability. These concepts can be defined as[14]:
- Durability: ability to cope with a chronic stress, where the source of this stress is endogenous to the system;
- Robustness: ability to recover or maintain the systems' social-ecological functions in the face of an external and chronic driver;
- Stability: system’s tolerance to transient and endogenous shocks or disruptions.
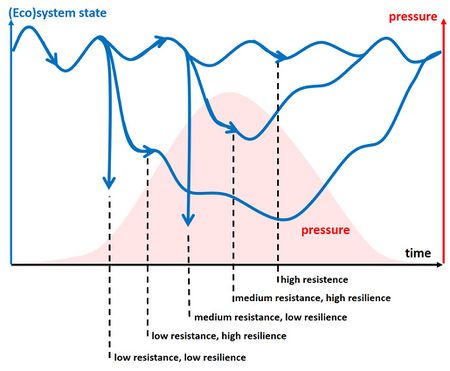
Both resistance and resilience cause an ecosystem to remain relatively unchanged when confronted to a disturbance, but in the case of resistance alone no internal re-organization and successional change is involved. This can lead to collapse of the system when a disturbance threshold is exceeded. In contrast, resilience implies that the system is internally re-organizing while retaining essentially the same function, structure, identity and feedbacks, perhaps through a mozaic of patches that are at different stages of re-assembly (Scheffer and Carpenter, 2003[15]). This self-organizing capacity prevents catastrophic collapse and allows for flexible adaptation to disturbance. The system may return after perturbation to its previous state but it may also shift to another ecosystem state. This behaviour can be analyzed in terms of nonlinear systems theory, that provides a mathematical framework for phenomena such as tipping points, bifurcations (regime shifts), hysteresis and multiple stable states (Nicolis and Prigogine, 1989[16]; Fisher and Pruitt, 2020[17]). Typical system responses to changing environmental conditions are displayed schematically in Figs. 1 and 2, corresponding to different resistance and resilience characteristics. See also the article Ecological thresholds and regime shifts. The behaviour sketched in these figures is strongly simplified. Resistance does not mean that an ecosystem is locked in a steady stable state. Due to the energy flow through the system (food, light, water motion), it moves cyclically or stochastically around stable states (so-called attractors), even without change in external environmental conditions.
When considering the potential effect of a certain type of disturbance it is thus useful to ask two questions:
- Will the species of this system be able to tolerate it (implying resistance), and if not,
- Is recovery possible through a successional trajectory, back to the same, or at least a desirable, ecosystem state (implying resilience)?
Resistance breaks down when uni-directional environmental change acts faster than the organisms' ability to adapt their tolerances. If uni-directional change is an ongoing process (for example climate change), the system will also lose resilience after a certain time, as full recovery through succession will then not be possible. Recovery from sudden and local disturbance is often possible through recolonization, but the rate of recovery will depend crucially on the spatial extent of disturbance. For example, recovery from anoxia could take 5 to 8 months at the scale of square meters (Rossi et al. 2009[18]), but could take 5 to 8 years at the scale of a whole bay (Diaz & Rosenberg, 1995[19]).
According to definition (4), the speed at which an ecosystem returns to its former state following a (minor) disturbance can be considered a measure of resilience. The idea is that a system with a short return time is more resilient than one with a long return time. Such resilience measured as (1 / the return time to a stable equilibrium) has also been called engineering resilience. It has however a long history of use among ecologists (Pimm 1982[20], DeAngelis 1992[21], Vos et al. 2005[22], Alexander 2013[23]). Resilience is also used in a way that more closely resembles the definition of resistance. Ecological resilience was defined as the amount of disturbance that an ecosystem could withstand without shifting to another ecological state characterized by another structure and other functions (definition 3).
Homeostasis
The word “homeostasis”, literally, indicates the absence of changes and an absolute maintenance of the status quo of an organism, but is generally understood as the coordinated physiological processes which maintain the steady functioning of the organism. Resistance and homeostasis are clearly related concepts. A homeostatic response to external perturbations may be selectively favored when the advantage offered by a reorganization of cell resources cannot be exploited. This is most likely to occur in the case of environmental perturbations of moderate intensity and short duration relative to the growth rate (Giordano, 2013[24]). However, some authors[23][14] define homeostasis as the self-regulation capacity of a system (organism) to resist or to adapt to external disturbances - a definition that includes both resistance and resilience.
Resilience and biodiversity
Resilience of coastal systems largely depends on biodiversity, which is a major requirement for allowing ecosystems to adapt to changing conditions (Loreau et al. 2001[25]). The human impact on the environment through pollution, fisheries, sediment erosion / deposition and global climate change has brought about much faster change than would occur under natural conditions, putting severe stress on many ecosystems (Luypaert et al. 2020[26]). Genetically diverse communities are more likely to contain a greater range of functional traits and environmental sensitivities. High diversity therefore entails opportunities for more efficient resource use as well as providing stability to ecosystem functions in variable environments and in the face of disturbance (Chapin et al. 1997[27]). Alternatively, systems with species-poor communities are theoretically likely to be functionally poorer, less resistant (capacity to resist change) and resilient (capacity to recover from change) to disturbance than systems with species-rich communities (Strong et al. 2015[28]) – see also the article Disturbances, biodiversity changes and ecosystem stability. Without genetic diversity, natural selection cannot occur and if natural selection is limited, adaptation is impossible. Preservation of biodiversity and, more specifically, genetic diversity is therefore of paramount importance for successful adaptation to our rapidly changing environments. According to present understanding, preservation of biodiversity does not necessarily imply conservation of the species community composition, because the maintenance of a particular species assemblage is not necessarily a requirement for the resilience of ecosystem functions (Gladstone-Gallagher et al. 2019[29]). It has also become clear that biodiversity may not always protect ecosystems from major abiotic disturbances (Folke et al. 2004[30]).
Resilience through recolonization
To understand resilience of ecosystems it is essential to understand what drives succession within ecosystems. Succession determines how, and how fast, communities return to their original state, or perhaps enter a new state. Many aspects of succession can be understood in terms of trade-offs between the ability to be either a good early (re)colonizer or a good competitor. Succession involves a gradual replacement of colonizer / competitor species according to the degree to which they tolerate, facilitate or inhibit certain environmental conditions and other species (Rossi et al. 2009[18]). The extent to which processes of (re)colonization and succession can take place largely determines the recovery of ecosystems after major disruption and is therefore an essential characteristic of the resilience of ecosystems (Gladstone-Gallagher et al. 2019[29]).
In this context, it is important to consider the spatial component of ecosystem resilience. Diversity of structurally and functionally connected landscapes, rich in resources and species, promotes the flow or movement of individuals, genes, and ecological processes. Below certain thresholds of connectivity the capacity to regain structure and function after perturbation is lost (Rappaport et al. 2015[31]). Chambers et al. (2019[32]), based on Allen et al. (2016[33]), have therefore introduced the concept of 'spatial resilience', which is a measure of how spatial attributes, processes, and feedbacks vary over space and time in response to disturbances and affect the resilience of ecosystems. Self-organization through strong feedbacks at multiple scales and high levels of functional diversity and redundancy, stabilizes the system with respect to disturbances within the range of historic variability.
When creating Marine Protected Areas, the sources of populations at all stages of succession should be protected, to preserve 'ecological memory' to the fullest possible extent. This includes protecting not only 'high quality' habitats that harbour healthy mature communities, but also 'low quality' and disturbed habitats that are required for those species that contribute to early recovery of perturbed areas (Rossi et al. 2009[18]). The selection of Marine Protected Areas thus involves evaluating the number, size, and spatial configuration of habitat fragments and degree of connectivity required to support restoration of ecosystems and conservation of focal habitats and species (Chambers et al. 2019[32]; Olsson et al. 2019[2]).
Resistance to long-term changes in abiotic and biotic factors
Community composition and ecosystem function may change very little under environmental change when the organisms can adapt to such change or tolerate it for some time (when the change is only temporary). However, all organisms have bounds to what they can temporarily or permanently tolerate, and when change exceeds some of these limits, the community composition and ecosystem functioning is likely to change.
It is unlikely that communities can be resistant to ongoing gradual change, such as global warming. Acclimation and phenotypic plasticity do not suffice to maintain the system as it is. Genetic adaptation could allow community members to track such abiotic environmental change, but it is more likely that the area where the community is functioning will be invaded by species that function well at higher temperatures. The original species will thus have to deal with new competitors and predators, in addition to the changed abiotic factor. To some extent the original community can track the preferred temperature range, by moving spatially to greater depths or to alternative geographic areas. But these new areas are likely to differ in other ecological aspects such as water pressure, light climate and perhaps speeds of water flow etc.
Adaptation and the consequences of mortality at different trophic levels
External disturbance interacts with internal mechanisms that shape community structure. To understand how an increased mortality of top-predators will affect the entire food chain, it is essential to understand how processes of mutual adaptation within food chains already give shape to existing patterns such as trophic structure (how biomass in ecosystems is partitioned between trophic levels) – see the article Trophic cascade.
Abundances at different trophic levels (such as algae, herbivores, carnivores and top-predators) and their responses to increased mortality (as under environmental change) depend critically on different mechanisms of adaptation within food chains and on the importance of population density at each of these trophic levels. However, different types of adaptation to living in a food chain context (balancing the need to acquire resources with the need to avoid predation) can often have similar consequences. For example, micro-evolution of behaviour, species replacement and induced defenses at a middle trophic level may all have similar effects on trophic level abundances in disturbed food chains (Abrams and Vos 2003[34]).
Assessment of resilience
The foregoing considerations can be summarized in a number of resilience characteristics (Walker et al. 2004[35]) – see also Fig. 1:
- Latitude: the maximum amount a system can be changed before losing its ability to recover within the same state (before crossing a threshold), represented by the width of the basin of attraction;
- Resistance: the ease or difficulty of changing the system, represented by the width of the resistance domain; deep basins of attraction indicate that greater disturbances are required to change the current state of the system;
- Precariousness: how close the current state of the system is to a limit or threshold that, if crossed, makes reorganisation difficult or impossible;
- Cross-scale interactions ('panarchy'): the resilience of a system at a particular spatial scale will depend on the influences from states and dynamics at spatial scales above and below.
Implications for ecosystems are (Gladstone-Gallagher et al. 2019[29]):
- For ongoing (chronic) changes and changes on large space scales (such as climate change), resilience is largely determined by the resistance of particular species: biological or behavioral traits that increase a species' resistance (tolerance) to disturbances. Resilience is likely best secured in genetically diverse and functionally redundant ecosystems: ecosystems in which many different species share the same combinations of functional (or resource use) characteristics.
- For short-term hefty disruptions, resilience is largely determined by spatial resilience (recolonization potential): the ability of populations to supply recruits to a disturbed patch and the ability of new recruits to colonize new space. Resilience is likely best secured in diverse connected ecological systems that are capable to change, adapt, and transform in response to stresses and strains through feedbacks across multiple spatial and temporal scales.
Related articles
- Ecological thresholds and regime shifts
- Disturbances, biodiversity changes and ecosystem stability
- Biodiversity and Ecosystem function
- Thresholds and Marine Policies
- Vulnerability and risk
- Species extinction
- Trophic cascade
- Integrated Coastal Zone Management (ICZM)
- Sustainability indicators
- Measurements of biodiversity
References
- ↑ 1.0 1.1 Lake, P.S. 2013. Resistance, Resilience and Restoration. Ecological Management and Restoration 14: 20-24
- ↑ 2.0 2.1 Olsson, S., Melvin, A. and Giles, S. (eds.) 2019. Climate change and ecosystems. Procs. Sackler Forum on Climate Change and Ecosystems, Washington, DC, November 8-9, 2018, organized by the National Academy of Sciences and The Royal Society
- ↑ Folke, C., Carpenter, S. R., Walker, B., Scheffer, M., Chapin, T. and Rockstrom, J. 2010. Resilience thinking: integrating resilience, adaptability and transformability. Ecology and Society 15(4): 20
- ↑ Scheffer, M. 2009. Critical transitions in nature and society. Princeton University Press, Princeton, New Jersey, USA
- ↑ 5.0 5.1 Brand, F.S. and K. Jax. 2007. Focusing the meaning(s) of resilience: resilience as a descriptive concept and a boundary object. Ecology and Society 12(1):23
- ↑ 6.0 6.1 6.2 Haines‐Young, R. and Potschin. M. (eds.) 2010. The Resilience of Ecosystems to Environmental Change (RECCE). Overview Report, 27 pp. Defra Project Code: NR0134
- ↑ Holling, C.S. 1973. Resilience and stability of ecological systems. Annual Rev. Ecol. Syst. 4: 1–23. doi: 10.1146/annurev.es.04.110173.000245
- ↑ Gunderson, L.H. 2000. Ecological Resilience - in Theory and Application. Annual Review of Ecology and Systematics 31:425-439.
- ↑ Hirota,M., Holmgren,M., Van Nes, E. H, and Scheffer,M. 2011. Global resilience of tropical forest and savanna to critical transitions. Science 334: 232–235. doi: 10.1126/science.1210657
- ↑ Chambers, J. C., Bradley, B. A., Brown, C. S., D’Antonio, C., Germino, M. J., Grace, J. B., et al. 2014. Resilience to stress and disturbance, and resistance to Bromus tectorum L. invasion in the cold desert shrublands of western North America. Ecosystems 7: 360–375. doi: 10.1007/s10021-013-9725-5
- ↑ Pope, K. L., Allen, C. R., and Angeler, D. G. 2014. Fishing for resilience. T. N. Am. Fisheries Soc. 143: 467–478. doi: 10.1080/00028487.2014.880735
- ↑ Seidl, R., Spies, T. A., Peterson, D. L., Stephens, S. L., and Hick, J. A. 2016. Searching for resilience: addressing the impacts of changing disturbance regimes on forest ecosystem services. J. Appl. Ecol. 53 : 120–129. doi: 10.1111/1365-2664.12511
- ↑ Ruggerio, C.A. 2021. Sustainability and sustainable development: A review of principles and definitions. Science of the Total Environment 786, 147481
- ↑ 14.0 14.1 14.2 Dawson, T.P., Rounsevell, M.D.A., Kluvankova‐Oravska, T., Chobotova V. and Stirling, A. 2010. Dynamic properties of complex adaptive ecosystems: implications for the sustainability of services provision. Biodiversity and Conservation 19: 2843‐2853
- ↑ Scheffer, M. and Carpenter, S. R. 2003. Catastrophic regime shifts in ecosystems: Linking theory to observation. Trends in Ecology and Evolution 18: 648–656
- ↑ Nicolis, G. and Prigogine, I. 1989. Exploring complexity: An introduction. New York, NY: W. H. Freeman. ISBN 0-7167-1859-6
- ↑ Fisher, D.N. and Pruitt, J.N. 2020. Insights from the study of complex systems for the ecology and evolution of animal populations. Current Zoology 66: 1–14
- ↑ 18.0 18.1 18.2 Rossi, F., Vos, M. & Middelburg, J.J. 2009. Species identity, diversity and microbial carbon flow in reassembling macrobenthic communities. Oikos 118: 503-512.
- ↑ Diaz, R.J. & Rosenberg R. 1995. Marine benthic hypoxia: a review of its ecological effects and the behavioural responses of benthic macrofauna. Oceanogr. Mar. Biol. Annu. Rev. 33:245-303.
- ↑ Pimm, S.L. 1982. Food Webs. The University of Chicago Press.
- ↑ DeAngelis, D.L. 1992. Dynamics of Nutrient Cycling and Food Webs. Chapman and Hall, London.
- ↑ Vos, M., Kooi, B.W., DeAngelis, D.L. & Mooij, W.M. 2005. Inducible defenses in food webs. In: Dynamic Food Webs. Multispecies Assemblages, Ecosystem Development and Environmental Change. Eds. P.C. de Ruiter, V. Wolters & J.C. Moore. Academic Press. Pp. 114-127
- ↑ 23.0 23.1 Alexander, D.E. 2013. Resilience and disaster risk reduction: an etymological journey. Nat. Hazards Earth Syst. Sci. 13: 2707–2716
- ↑ Giordano, M. 2013. Homeostasis: An underestimated focal point of ecology and evolution. Plant Science 211: 92– 101
- ↑ Loreau, M., Naeem, S., Inchausti, P., Bengtsson, J., Grime, J. P., Hector, A., Hooper, D. U., Huston, M. A. ,Raffaelli, D., Schmid, B., Tilman, D. and Wardle D. A. 2001. Biodiversity and Ecosystem Functioning: Current Knowledge and Future Challenges. Science 294: 804-808
- ↑ Luypaert T., Hagan J.G., McCarthy M.L., Poti M. 2020. Status of Marine Biodiversity in the Anthropocene. In: Jungblut S., Liebich V., Bode-Dalby M. (eds) YOUMARES 9 - The Oceans: Our Research, Our Future. Springer, Cham. https://doi.org/10.1007/978-3-030-20389-4_4
- ↑ Chapin, F.S., Walker, B.H., Hobbs, R.J., Hooper, D.U., Lawton, J.H., Sala, O.E. and Tilman D. 1997. Biotic control over the functioning of ecosystems. Science, 277(5325): 500-504
- ↑ Strong, J.A., Andonegi, E., Bizsel. K.C., Danovaro, R., Elliott, M., Franco, A., Garces, E., Little, S.,. Mazik, K., Moncheva, S., Papadopoulou, N., Patrício, J., Queiros, A.M., Smith, C., Stefanova, K. and Solaun, O. 2015. Marine biodiversity and ecosystem function relationships: The potential for practical monitoring applications. Estuarine, Coastal and Shelf Science 161: 46-64
- ↑ 29.0 29.1 29.2 Gladstone-Gallagher, R.V., Pilditch, C.A., Stephenson, F. and Thrush, S.F. 2019. Linking Traits across Ecological Scales Determines Functional Resilience. Trends in Ecology and Evolution 34 (12) https://doi.org/10.1016/j.tree.2019.07.010
- ↑ Folke, C., Carpenter, S., Walker, B., Scheffer, M., Elmqvist, T., Gunderson, L. & Holling, C.S. 2004. Regime Shifts, Resilience, and Biodiversity in Ecosystem Management. Annual Review of Ecology and Systematics 35:557-581.
- ↑ Rappaport, D. I., Tambosi, L. R. and Metzger, J. P. 2015. A landscape triage approach: combining spatial and temporal dynamics to prioritize restoration and conservation. J. Appl. Ecol. 52: 590–601
- ↑ 32.0 32.1 Chambers, J.C., Allen, C.R. and Cushman, S.A. 2019. Operationalizing Ecological Resilience Concepts for Managing Species and Ecosystems at Risk. Front. Ecol. Evol. 7:241. doi: 10.3389/fevo.2019.00241
- ↑ Allen, C. R., Angeler, D. G., Cumming, G. S., Folk, C., Twidwell, D., and Uden, D. R. 2016. Quantifying spatial resilience. J. Appl. Ecol. 53, 625–635. doi: 10.1111/1365-2664.12634
- ↑ Abrams, P.A & Vos, M. 2003. Adaptation, density dependence and the responses of trophic level abundances to mortality. Evolutionary Ecology Research 5: 1113-1132
- ↑ Walker, B., Holling, C.S., Carpenter, S.R. and Kinzig, A. 2004. Resilience, adaptability and transformability in social – ecological systems. Ecology and Society 9(2): 5. http://www.ecologyandsociety.org/vol9/iss2/art5
Please note that others may also have edited the contents of this article.
|